765
Views & Citations10
Likes & Shares
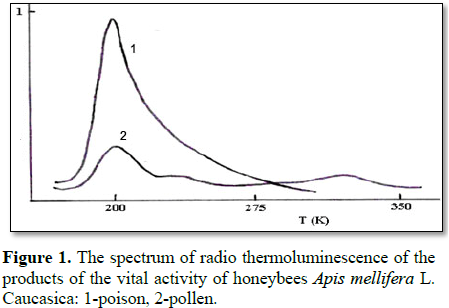
The presented work presents data on study of radiation effects on bee venom using of methods radiothermoluminescence. During analysis, the samples were irradiated at 77 K and a dose of 0.2×104 Gy, while the heating rate when receiving the emission spectrum was 2.5 and 5.8 deg/min. Experimentally it was found that the radio-luminescence curve with a maximum at 200 K is typical for bee venom, and for pollen there is a maximum emission at 190 K, 240 K and a weak peak at 337 K.
As a result of the studies conducted, g-radiation doses (D=0.5, 1.0, 1.5 and 2.5 kGy) were selected for radiation sterilization of bee venom and preparations based on them. The obtained data can be recommended to the pharmaceutical industry for the radiation sterilization of poison and preparations based on bee venom in order to increase their shelf life.
Keywords: Honey bee, Venom, Pollen, Radiothermoluminescence, Radiation sterilization
INTRODUCTION
Among a huge number of biologically active substances of
natural origin, one of the central places is occupied by animal poisons - a
group of compounds unique in chemical nature and physiological action. Toxic
and medicinal properties of them are known to man since ancient times. It
should be noted that while some insects have more than 1.000.000 species, there
are over 300.000 plant species in the world at present, Exceeding the plants in
the number of species, the animals are incomparably poorer in their number of
individuals, which probably explains the small study of the pharmacological and
biochemical properties of biologically active substances of animal origin. From
ancient times human beings, coming into contact with wildlife, came across
various poisonous animals. Often, such a collision with poisonous insects,
amphibians and reptiles led to the death of a person. At present, many
biologically active substances of animal origin have been identified and their
biochemical, physicochemical and pharmacological properties have been studied.
The Manual on International Statistical Classification
of Diseases, Trauma and Causes of Death (WHO, Geneva) includes poisoning and
toxic reactions due to exposure to poisonous plants and animals, including code
E905.3 Bites of hornets, wasps, bees. According to scientific data, bees
existed for 56 million years before the appearance of primitive man [1].
The honey bee (Apis
mellifera L.) is a part of the superfamily of Apoidea, which forms a large
group (about 20% of the species composition) in the progressing order of
Hymenoptera [2].
Animal venoms and toxins consist of proteins and
peptides with potential medical and biotechnological use. Apitoxins are
produced and secreted through a gland (acid gland) located in the posterior
region of the Apis mellífera bee’s abdomen, containing a complex mixture of
substances with
The venom proteome of the honeybee A. mellifera was recently investigated
by integrating a combinatorial peptide ligand library approach with nanoLC
FT-ICR MS/MS [11], resulting in 102 venom proteins and peptides, of
which 33 were categorized as putative venom toxins.
The quantity and quality of bee venom depends on the
age of the bees, forage quality and season. Bee venom contains trace elements
phosphorus, copper, calcium, magnesium and other elements. Bee venom is
composed of 18 amino acids. Enzyme action of bee venom is 30 times more potent
than the snake. Its activity maintains for 7-8 years. No visible effect
withstands freezing and heating to 110-115°C. Bee venom is resistant to the
effects of acids and alkalis. Lead peptide in bee venom melittin is composed of
26 amino acids (50-55% dry matter venom) [12-15].
The study of the influence of environmental factors
on living organisms, including on the products of their vital activity, the
study of biochemical, physicochemical, pharmacological and toxicological
properties of them is of great importance in the development of conditions for
radiation sterilization of both venom and preparations based on them, which is
the basis of the concept and the strategy of rational use of bioresources, one
of the components of which in Azerbaijan is the honey bee.
Analyzing literary data, it is necessary to note
that despite the thorough research of the products of vital activity of honey
bees, there is a need for more in depth and comprehensive study of the products
of their vital activity. This problem still does not lose its relevance.
Equally important is the increase in the shelf life
of poison and other life products of the honey bee.
Based on the above, the purpose of the research was
to identify doses of gamma radiation for the radiation sterilization of the
products of life products of the honey bee.
MATERIALS AND METHODS
Experiments on the study of the effect of g-radiation on the chemical composition
of the products under study were carried out by the method of radio thermal
luminescence. Irradiation and the effect of radiation on poison and pollen were
performed on a K-25 isotope plant using 60Co.
We have carried out experimental studies on the
study and identification of the effects of low doses of gamma radiation on
molecular mobility and changes in the characteristic temperatures of the poison
and pollen.
As a result of experimental work, comparative
studies were carried out on the effect of low doses of g-radiation up to 104
Gy of exposure in air on molecular mobility, changes in characteristic
temperatures, spectral characteristics and pharmacological properties of Apis mellifera L. honey bee products
(poison, pollen), using radiothermoluminescence.
Gamma irradiation of the samples of poison and
pollen in the form of tablets pressed at room temperature was carried out both
in vacuum and in air from a source of 60Co. The test samples in an amount of
0.2 mg were placed in metal cuvettes for irradiation with a gamma-ray source.
Samples were irradiated with small doses of g-radiation. On the basis of 5-fold measurements, their
radio-thermoluminescence spectra were constructed.
THE RESULTS OF THE STUDY AND DISCUSSION
In radio thermoluminescent analysis, the samples
were irradiated at 77 K and a dose of 0.2×104 Gy, while the heating rate upon receipt of the emission
spectrum was 2.5 and 5.8 deg/min.
It is shown that for samples of bee venom, a
characteristic of radio thermoluminescence with a maximum at 200 K is
characteristic. The intensity of the glow from 200 K exponentially decreases to
300 K.
In contrast to the honeybee venom, for pollen,
luminescence peaks are observed at 190 K, 240 K and a weak peak at 337 K. The
nature of these peaks is currently being clarified. It is only established that
the intensity and temperature position of the first peak do not depend on the
heating rate. Irradiation in air leads to an increase in the intensity of
luminescence of low-temperature peaks, which can be associated with the
influence of dissolved oxygen O2 in the samples under study.
Many questions about
the effect of low doses of g-radiation, and other types of ionizing radiation on a living organism
remain open. These questions are important for the technology of radiation
sterilization of drugs.
The literature
provides data on the study of bee venom, but many questions still remain
unanswered and require in-depth analysis and study.
We used the method of
radio-thermoluminescence to study the effects of radiation on bee venom, which
allowed us to obtain information about the structural properties of the system,
about the centers of charge stabilization of the primary products of the
radiolysis of the poison, about the migration pathways absorbed during
irradiation and so on.
Poison samples were
irradiated in a special cuvette with gamma rays at 77 K to doses of % 5 kGy.
Before irradiation, the samples were cleaned from traces of oxygen. Irradiation
was carried out in air and in vacuum. The luminescence curves were recorded at
a rate of ~ 5°/min.
Studies conducted by
us have shown that with an increase in the dose of radiation in solutions of a
poison, there is a significant change in the optical density of the samples. In
this case, there is a decrease in the intensity of absorption at 200 nm, which
is a result of the occurrence of biochemical reactions in the solid phase of
individual zootoxin enzymes.
Comparison of the
intensity of absorption of non-irradiated samples of poison with samples of
zootoxin irradiated to doses of 2.5 kGy did not reveal significant changes.
However, it was found
that exposure to g-radiation at doses of D=3.5, 4.0, 4.5 and 5.5 kGy for 3 min showed a
decrease in toxicity of the poison. We assume that this is the result of a
change in the pharmacological activity of the enzymes of the poison.
Thus, g-radiation doses (D=0.5,
1.0, 1.5 and 2.5 kGy) are recommended for radiation sterilization of poison and
preparations based on them.
Summarizing the
experimental results obtained, it can be concluded that, under the influence of
g-radiation (up to doses of
2.5 kGy) on the poison per 3 min, there is no reduction in the intensity of
absorption and accordingly, toxicity, including pharmacological activity.
A further increase in
the dose of g-radiation in the range from 3.5 to 5.5 kGy leads to a gradual decrease
in both toxicity and pharmacological activity of the poison.
We assume that the
effect of g-radiation (up to doses of 2.5 kGy) on a solution of bee venom over a
period of 3 min helps stabilize the toxicity and pharmacological activity of
the poison. Undoubtedly, the stabilization of the pharmacological activity of
the poison will lead to an increase in the shelf life of aqueous solutions of
the poison and preparations based on the poison of the bees.
Therefore,
experimentally proven figures (up to doses of 2.5 kGy and irradiation time of 3
min) can be used to sterilize aqueous solutions of bee venom.
CONCLUSION
It was revealed that
radio-thermoluminescence with a maximum at 200 K is typical for poison and for
pollen - maximum of luminescence at 190 K, 240 K and a weak peak at 337 K.
Recommended doses
of-radiation (D=0.5, 1.0, 1.5 and 2.5 kGy) for radiation sterilization of poison
and preparations based on them.
The obtained data can
be recommended to the pharmaceutical industry for radiation sterilization of
poison and their preparations in order to increase the shelf life.
- http://www.docs.cntd.ru/document/420317970
- https://www.scienceforum.ru/2017/2263/28551
- Oršolić N (2012) Bee venom in cancer therapy. Cancer Metastasis Rev 3: 173-194.
- Danneels EL (2015) Honeybee venom proteome profile of queens and winter bees as determined by a mass spectrometric approach. Toxins 7: 4468-4483.
- Sobral F (2016) Chemical characterization, antioxidant, anti-inflammatory and cytotoxic properties of bee venom collected in Northeast Portugal. Food Chem Toxicol 94: 172-177.
- Sciani JM (2010) Identification of a novel melittini so form from Africanized Apis mellifera venom. Peptides 31: 1473-1479.
- Ferreira Junior RS (2010) Africanized honey bee (Apis mellifera) venom profiling: Seasonal variation of melittin and phospholipase A2 levels. Toxicon 56: 355-362.
- Han SM (2016) Evaluation of anti-acne property of purified bee venom serum in humans. J Cosmetic Dermatol 15: 1-6.
- Lee S (2016) Antifungal activity of bee venom and sweet bee venom against clinically isolated Candida albicans. J Pharmacopuncture 19: 45-50.
- Gajski G (2013) Melittin: A lytic peptide with anticancer properties. Environ Toxicol Pharmacol 36: 697-705.
- Lee WR, Kim KH, An HJ, Kim JY, et al. (2014) The protective effects of melittin on Propionibacterium acnes induced inflammatory responses in vitro and in vivo. J Investig Dermatol 134: 1922-1930.
- Abrantes AF, da Rocha TC, de Lima AB, Cavalcanti MT (2017) Honey bee venom: Influence of collection on quality and cytotoxicity. Ciênc Rural 47: e20160486.
- Topchiyeva S, Mehrabova MA, Abiyev HA (2009) Investigation of electrophysical parameters of snake venom. The International Conference on Structural Analysis of Advanced Materials 1: 57-58.
- Krylov VN (1993) Bee venom - what is it. Beekeeping, pp: 36-39.
- Krylov V, Oshevensky L, Kurnikov G, Klemenova I (1999) Cream based on wax with propolis and poyal jelly in the treatment of dermatological diseases. Apimondia’99. Congress Vancouver, Canada. Proceedings 36: 249.